Course info
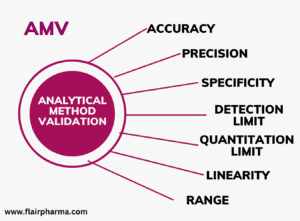
This course offers a comprehensive exploration of advanced concepts in pharmaceutical quality control and analytical method validation, emphasizing both theoretical principles and practical applications. It covers the design, development, and validation of analytical methods in line with international regulatory standards (ICH, WHO, FDA), ensuring the quality, safety, and efficacy of pharmaceutical products. Key topics include advanced quality control strategies, modern analytical techniques, method validation parameters (accuracy, precision, specificity, linearity, robustness), statistical evaluation, and regulatory documentation. The course also addresses cGMP compliance, regulatory inspections, and the critical interface between Quality Control (QC) and Quality Assurance (QA). Through case studies and applied exercises, learners will develop the skills to implement and critically assess analytical methods in real-world pharmaceutical settings.
- Teacher: Samira Karim